よむ、つかう、まなぶ。
資料3-4 研究報告の報告状況[920KB] (29 ページ)
出典
| 公開元URL | https://www.mhlw.go.jp/stf/newpage_70725.html |
| 出典情報 | 薬事審議会 医薬品等安全対策部会(令和7年度第4回 3/6)《厚生労働省》 |
ページ画像
ダウンロードした画像を利用する際は「出典情報」を明記してください。
低解像度画像をダウンロード
プレーンテキスト
資料テキストはコンピュータによる自動処理で生成されており、完全に資料と一致しない場合があります。
テキストをコピーしてご利用いただく際は資料と付け合わせてご確認ください。
研究報告の報告状況
(2025年8月1日~2025年11月30日)
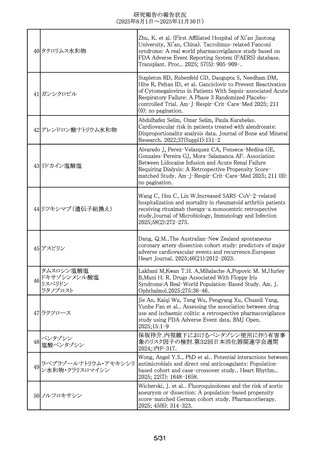
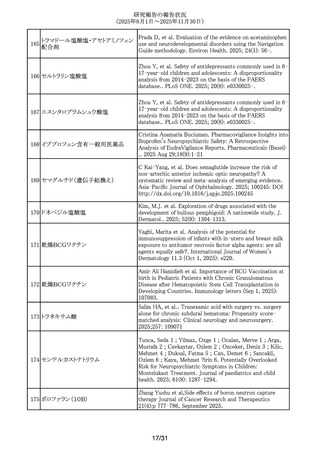
ビフィズス菌
ラクトミン
ビフィズス菌含有一般用医薬品
291
耐性乳酸菌製剤(3)含有一般用医薬
品
ラクトミン含有一般用医薬品
292 レベチラセタム
293 バルプロ酸ナトリウム
294 バルプロ酸ナトリウム
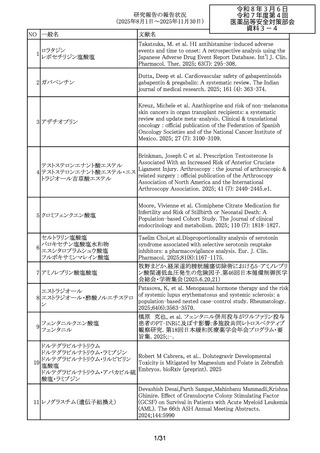
Munefumi Shimosaka,Yudai Ishida,Shinji Takamatsu,他.
Oral administration of Enterococcus faecalis to mice induces
acute and chronic inflammation in the pancreas: possible
involvement in the pathophysiology of pancreatic diseases.
Biochemical and Biophysical Research Communications 790
(2025) 152880
Li J, et al. Psychiatric disorders associated with newer
antiseizure medications: A real-world disproportionality
analysis of FDA adverse event reporting system. Epilepsy
Behav. 2025; 172: 110722-.
Paternal Valproate Use and Neurodevelopmental Disorder
and Congenital Malformation Risk in Offspring. JAMA
network open 2025; 8(11): e2542581
Paternal valproate use and impact of shared genetic
susceptibility on child neurodevelopment. Scientific reports
2025; 15(1): 39033.
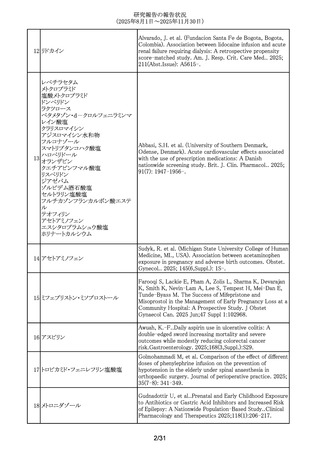
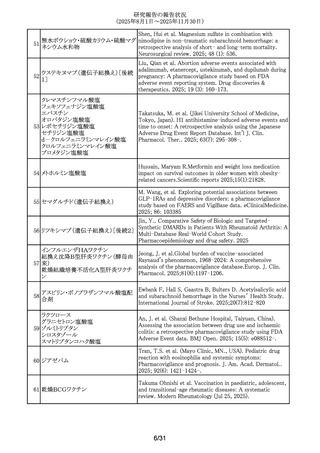
295 フェノバルビタール
Mazzone, P.P.Association of maternal epilepsy with perinatal
outcomes, and an exploration of prenatal antiseizure
medication: A population-based retrospective cohort
study.Epilepsia. 2025;66(9):3353-3368.
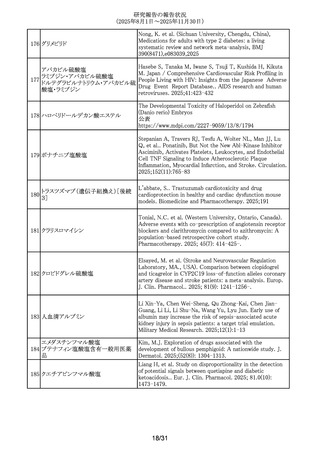
296 プロプラノロール塩酸塩
Watt, A. et al. Poisoning in adolescents in the UK: a review
of enquiries to the National Poisons Information Service.
Arch.Dis.Child. 110(9),687-692,2025.
297 アバコパン
Yang, Zhenyao; Wang, Chengzhi; Wei, Shiju; Liu, Yan; He,
Mingyang; Zheng, Chenglei; Li, Wenjun; Li, Dongdong; Liu,
Peimin. Post-marketing safety profile and clinical
prioritization of adverse events with avacopan in ANCAassociated vasculitis: a FAERS pharmacovigilance analysis.
Renal failure. 2025; 47(1): -2568971.
298 アムロジピンベシル酸塩
Kappelin, J. et al. Antihypertensive medication as a risk
factor for basal cell carcinoma: A nationwide registry-based
case-control study. Acta Dermato-Vener. 2025.
105,adv42749
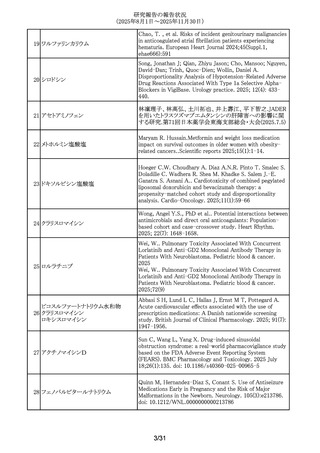
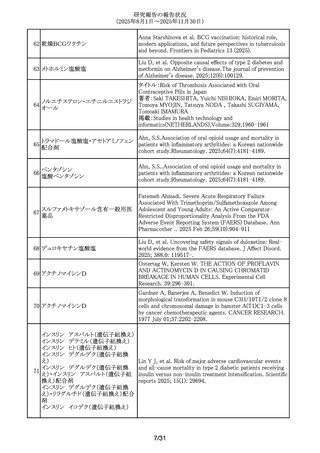
299 アバコパン
Li, Yang; Meng, Anna; Wang, Yue. The safety profiles of
avacopan on microscopic polyangiitis and granulomatosis
with polyangiitis: a real-world pharmacovigilance analysis.
Frontiers in immunology. 2025; 16: -1654735.
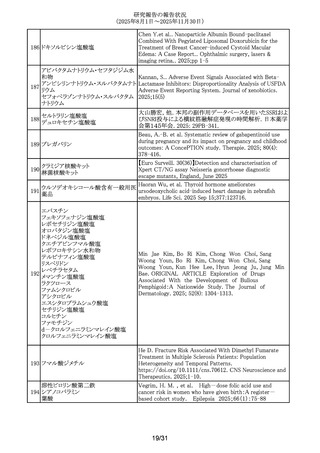
300 レベチラセタム
Mazzone, P.P. et al. (University of Edinburgh, Edinburgh,
UK). Association of maternal epilepsy with perinatal
outcomes, and an exploration of prenatal antiseizure
medication: A population-based retrospective cohort study.
Epilepsia. 2025; 66(9): 3353-3368-.
301 アムロジピンベシル酸塩
Kappelin, J. et al. (Lund University, Lund, Sweden).
Antihypertensive medication as a risk factor for basal cell
carcinoma: A nationwide registry-based case-control study.
Acta Dermato-Vener.. 2025; 105: adv42749-.
29/31
(2025年8月1日~2025年11月30日)
ビフィズス菌
ラクトミン
ビフィズス菌含有一般用医薬品
291
耐性乳酸菌製剤(3)含有一般用医薬
品
ラクトミン含有一般用医薬品
292 レベチラセタム
293 バルプロ酸ナトリウム
294 バルプロ酸ナトリウム
Munefumi Shimosaka,Yudai Ishida,Shinji Takamatsu,他.
Oral administration of Enterococcus faecalis to mice induces
acute and chronic inflammation in the pancreas: possible
involvement in the pathophysiology of pancreatic diseases.
Biochemical and Biophysical Research Communications 790
(2025) 152880
Li J, et al. Psychiatric disorders associated with newer
antiseizure medications: A real-world disproportionality
analysis of FDA adverse event reporting system. Epilepsy
Behav. 2025; 172: 110722-.
Paternal Valproate Use and Neurodevelopmental Disorder
and Congenital Malformation Risk in Offspring. JAMA
network open 2025; 8(11): e2542581
Paternal valproate use and impact of shared genetic
susceptibility on child neurodevelopment. Scientific reports
2025; 15(1): 39033.
295 フェノバルビタール
Mazzone, P.P.Association of maternal epilepsy with perinatal
outcomes, and an exploration of prenatal antiseizure
medication: A population-based retrospective cohort
study.Epilepsia. 2025;66(9):3353-3368.
296 プロプラノロール塩酸塩
Watt, A. et al. Poisoning in adolescents in the UK: a review
of enquiries to the National Poisons Information Service.
Arch.Dis.Child. 110(9),687-692,2025.
297 アバコパン
Yang, Zhenyao; Wang, Chengzhi; Wei, Shiju; Liu, Yan; He,
Mingyang; Zheng, Chenglei; Li, Wenjun; Li, Dongdong; Liu,
Peimin. Post-marketing safety profile and clinical
prioritization of adverse events with avacopan in ANCAassociated vasculitis: a FAERS pharmacovigilance analysis.
Renal failure. 2025; 47(1): -2568971.
298 アムロジピンベシル酸塩
Kappelin, J. et al. Antihypertensive medication as a risk
factor for basal cell carcinoma: A nationwide registry-based
case-control study. Acta Dermato-Vener. 2025.
105,adv42749
299 アバコパン
Li, Yang; Meng, Anna; Wang, Yue. The safety profiles of
avacopan on microscopic polyangiitis and granulomatosis
with polyangiitis: a real-world pharmacovigilance analysis.
Frontiers in immunology. 2025; 16: -1654735.
300 レベチラセタム
Mazzone, P.P. et al. (University of Edinburgh, Edinburgh,
UK). Association of maternal epilepsy with perinatal
outcomes, and an exploration of prenatal antiseizure
medication: A population-based retrospective cohort study.
Epilepsia. 2025; 66(9): 3353-3368-.
301 アムロジピンベシル酸塩
Kappelin, J. et al. (Lund University, Lund, Sweden).
Antihypertensive medication as a risk factor for basal cell
carcinoma: A nationwide registry-based case-control study.
Acta Dermato-Vener.. 2025; 105: adv42749-.
29/31